- Blog
- World war z pc game buy
- Embriologia clinica moore pdf gratis
- Matlab 2018b system requirements
- Combos kof 2002
- Breath of the wild rom for cemu
- Prasanth letter
- Tri state trucking
- Jagannatha hora bhava chakra
- Juno meaning
- Osho ashram
- Digi 003 plus
- Martensite start using jmatpro
- Top 10 video makers
- Volbeat still counting correct version clone hero
- Games like pubg for pc
- Maladolescenza porn
- Astrill vpn vip china
- Chinkee tan free ebook
- Kabhi alvida naa kehna images
- Amd radeon vega 3
- Stranger things season 1 episode 3
- Dua qunoot arabic
- Planswift 10 tutorial
- Pablo escobar the drug lord ep 2
- Free excel add ins
- Britney spears gimme more ti download
- Watch oz tv show
- The 10x rule rule pdf
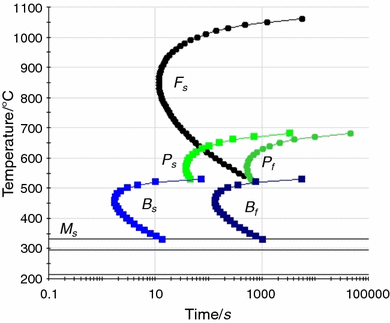
It is worth noting that Andrews equation does not take into account the amount of silicon in the steel, which is significantly higher in material B.
Martensite start using jmatpro software#
While the martensite start temperature for material A and C (determined using JmatPro software and Andrews equation) was comparable, the results for material B were inconsistent. The martensite start temperature established from the CCT curves and those determined from Andrews equation are summarised in Table 4. Figure 7 shows the CCTs for the three materials that were determined using JmatPro software. Despite the slightly smaller weight, the oxidation rate of material C is less than that of A, but higher than B. The oxidation rates were calculated when the graph stabilised. Material A showed a higher oxidation rate, whilst material B showed itself to be more resistant to oxidation.

The oxidation rate was determined by plotting the gradient once the graph on Fig. Figure 5 shows the weight gain due to heating and exposure to the atmosphere. Table 3 presents the sample weight before analysis and the weight after heating and exposure to the atmosphere. The graph for material C fell between that for material A and B. Figure 4 shows the behaviour of the three materials during heating, whilst purging with argon, followed by isothermal treatment for 60 min in the atmosphere. The trends of the graphs show that material C requires significantly higher heat flow to achieve the eutectoid temperature at approximately the same time as material A and B. The martensite start temperature was estimated using JMatPro practical software and compared with the Andrews equation 1.įigure 3 presents the required heat flow over time. Continuous cooling temperature (CCT) curves were plotted using the software. JMatPro software was used to predict the thermal properties of the materials. Initially, steel rods are heated in a controlled furnace to approximately \(850\,\) for 60 min under atmospheric conditions. Generally, the heat treatment cycle involved in the spring manufacturing process has the following sequence: In order for the heat treatment process to be optimised, the precise parameters have to be determined. The heat treatment process entails phase development when heated and cooled at determined rates. This will assist with optimisation and increasing efficiency during the heat treatment process. It is important to know the exact transformation temperatures, in order to determine the maximum heat treatment efficiency and the effect thereof on the structure of the specific spring steel. During the heat-treating processes used with spring steel, phase transformations occur that change the crystalline structure and affect the mechanical properties of the steel. Material A showed a high oxidation rate with decreased decarburisation, whereas material B showed less oxidation and higher decarburisation due to higher silicon contents.īogie springs are manufactured from high strength steels that have to be heat-treated properly, in order to produce functional springs properties. The three materials analysed showed distinct properties therefore, understanding of the material characteristics is important. The results of these investigations showed that there is a nonlinear relationship between oxidation and decarburisation. This allowed for the observation of phase transition and oxidation rate for each steel.

The materials were also subjected to a regular spring steel heat treatment process in order to determine the relationship between oxidation rate and decarburisation. The change in the corresponding heat flow was evaluated. Three spring steels with known mass were heated and cooled. The oxidation rates and material-specific phase transformation of the spring steels were evaluated by means of differential scanning calorimetry. Oxidation and decarburisation can adversely affect the surface properties of steel when not limited to the bare minimum. During the heat treatment of springs for railway bogies, phase transformations and oxidation occur and it is important to know transformation temperatures and oxidation rates to optimise the process.
- Blog
- World war z pc game buy
- Embriologia clinica moore pdf gratis
- Matlab 2018b system requirements
- Combos kof 2002
- Breath of the wild rom for cemu
- Prasanth letter
- Tri state trucking
- Jagannatha hora bhava chakra
- Juno meaning
- Osho ashram
- Digi 003 plus
- Martensite start using jmatpro
- Top 10 video makers
- Volbeat still counting correct version clone hero
- Games like pubg for pc
- Maladolescenza porn
- Astrill vpn vip china
- Chinkee tan free ebook
- Kabhi alvida naa kehna images
- Amd radeon vega 3
- Stranger things season 1 episode 3
- Dua qunoot arabic
- Planswift 10 tutorial
- Pablo escobar the drug lord ep 2
- Free excel add ins
- Britney spears gimme more ti download
- Watch oz tv show
- The 10x rule rule pdf
